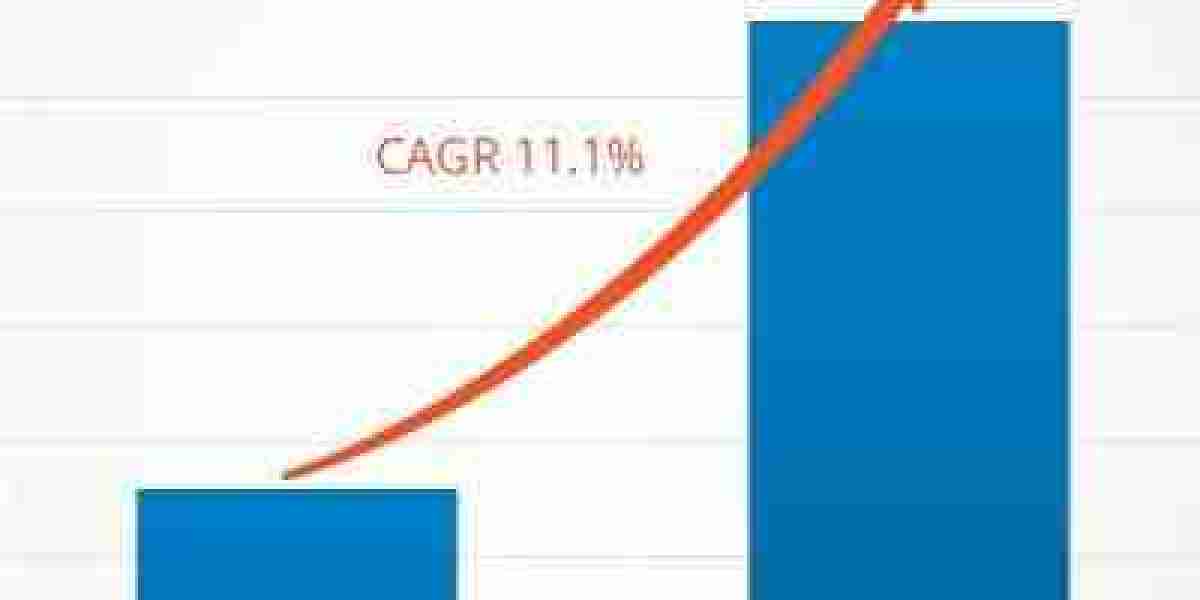
The global Medical Writing Market is experiencing strong expansion as pharmaceutical, biotechnology, and medical device companies increasingly rely on precise scientific documentation for regulatory submissions, clinical research, and product approvals. A detailed Medical Writing Market News and Recent Developments landscape highlights accelerating adoption of AI-enabled writing tools, rising outsourcing to CROs, expansion of decentralized clinical trials, and increasing regulatory updates from agencies such as the FDA and EMA, all of which are reshaping industry workflows and boosting demand for skilled medical writers. According to market estimates, the sector is projected to grow from US$ 3,931.50 million in 2023 to US$ 9,114.91 million by 2031, registering a strong CAGR of 11.1% during 2023–2031, driven by increasing complexity in clinical documentation and global regulatory compliance requirements.
Medical writing is a critical function across the drug development lifecycle, enabling the creation of clinical study reports, protocols, investigator brochures, safety summaries, and scientific publications. As pharmaceutical innovation accelerates in high-growth therapeutic areas such as oncology, rare diseases, gene therapies, and biologics, the need for structured, accurate, and compliant documentation continues to rise. This growing complexity is further reinforced by evolving global regulatory expectations and the need for consistent scientific communication across multiple geographies.
Download Sample PDF Report: https://www.theinsightpartners.com/sample/TIPRE00008502
Market News and Recent Developments: Industry Transformation
The Market News and Recent Developments in the Medical Writing Market indicate a rapid shift toward digital transformation and automation. Leading contract research organizations (CROs) and pharmaceutical companies are increasingly deploying generative AI and advanced document automation tools to streamline clinical documentation workflows, reduce turnaround times, and improve consistency in regulatory submissions. However, industry insights suggest that while AI enhances efficiency, human expertise remains essential for ensuring scientific accuracy and compliance.
Another significant development is the growing adoption of decentralized clinical trials (DCTs). These trials rely heavily on remote patient data collection, telehealth integration, and real-world evidence, which has increased the complexity of documentation requirements. As a result, medical writers are now required to manage diverse data sources and ensure regulatory-grade reporting across hybrid clinical models.
Recent industry movements also highlight a strong increase in outsourcing activities, with pharmaceutical and biotech companies shifting medical writing tasks to specialized service providers. This trend is driven by cost optimization strategies, the need for specialized expertise, and faster regulatory submission timelines. CROs are expanding global delivery centers, particularly in cost-efficient regions such as Asia-Pacific and Eastern Europe, to meet rising demand.
In addition, regulatory updates and evolving guidelines from authorities such as the FDA, EMA, and ICH are continuously shaping documentation standards. Frequent revisions to clinical trial reporting formats and pharmacovigilance requirements are increasing the need for experienced medical writers capable of adapting to dynamic regulatory frameworks.
The market is also witnessing a surge in clinical trial activity and biologics development, which is creating additional demand for medical documentation services. The rise in orphan drug approvals, personalized medicine, and advanced therapies such as cell and gene therapy is further expanding the scope of medical writing services globally.
Market Drivers Supporting Growth
One of the key drivers of the Medical Writing Market is the increasing number of global clinical trials, which require extensive and standardized documentation. The growing complexity of multi-regional studies is further intensifying demand for professional medical writing services.
Strict regulatory requirements also play a crucial role in market expansion. Regulatory agencies mandate detailed and accurate documentation for drug approvals, encouraging pharmaceutical companies to invest in specialized writing services.
The rising trend of outsourcing continues to strengthen market growth, as organizations seek efficiency, cost reduction, and access to domain expertise through external service providers.
Emerging Trends and Opportunities
The integration of AI, cloud-based platforms, and digital collaboration tools is improving efficiency in medical writing workflows, enabling faster document creation and review cycles. Additionally, the increasing use of real-world evidence (RWE) is expanding documentation requirements beyond traditional clinical trials.
The shift toward patient-centric communication is also driving demand for simplified medical content, including patient education materials and digital health communication tools.
Leading Market Players
Key companies operating in the global Medical Writing Market include:
- ICON plc
- Parexel International Corporation
- IQVIA Inc.
- Syneos Health
- Certara Inc.
- Labcorp Drug Development (Covance)
- Freyr Solutions
- Trilogy Writing & Consulting GmbH
- Caidya
- BioScience Writers
These players are actively investing in digital transformation, AI integration, and global expansion strategies to strengthen their market presence.
Future Outlook
The Medical Writing Market is poised for strong and sustained growth through 2031, driven by rising clinical trial complexity, regulatory changes, outsourcing trends, and rapid technological advancements. With increasing Market News and Recent Developments centered on AI adoption and decentralized trials, the industry is evolving into a highly dynamic and technology-driven ecosystem, supporting global pharmaceutical innovation and compliance.
About Us
The Insight Partners is a one stop industry research provider of actionable intelligence. We help our clients in getting solutions to their research requirements through our syndicated and consulting research services. We specialize in industries such as Semiconductor and Electronics, Aerospace and Defense, Automotive and Transportation, Biotechnology, Healthcare IT, Manufacturing and Construction, Medical Device, Technology, Media and Telecommunications, Chemicals and Materials.
Contact Us
Ankit Mathur | The Insight Partners
E-mail: [email protected]
Phone: +1-646-491-9876
Also Available in : Korean | German | Japanese | French | Chinese | Italian | Spanish